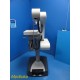
Carl Zeiss INTRABEAM NC32 I...
Carl Zeiss INTRABEAM NC32 IORT System w/ XRS4 PRS500 UNIDOS E Dosimetry ~ 39155
Item specifics
- Used
- Model
- NC32 INTRABEAM
MPN- 000000-1096-945
Product details
Manufacturer | Carl Zeiss Meditec AG |
Item: | Carl Zeiss INTRABEAM NC32 IORT System w/ XRS4 PRS500 UNIDOS E Dosimetry & Docs |
Model/Cat # | NC32 INTRABEAM | Ref 000000-1096-945 |
Serial No. | 6630467666 |
Made In: | Germany |
Date of Manufacture: | June 2016 |
Voltage: 100 / 115 / 230 V | 50–60 Hz | 400 VA
Short Description:
Carl Zeiss INTRABEAM® intraoperative radiation therapy (IORT) system designed for delivery of low-energy targeted radiation during surgical procedures.
System includes the NC32 mobile treatment arm, PRS 500 control console, PRS 500 user terminal workstation, XRS 4 50kV x-ray source, PTW UNIDOS E electrometer, PTW TN23342A ionization chamber, photodiode detector array, QA tools, sterile containers, cables, and extensive documentation.
System also includes quality assurance measurement equipment and historical calibration documentation, which can assist with recommissioning and physics validation after installation.
Spherical applicators are NOT included in this sale.
Fuse Ratings:
- 230–240V: T3.15A / 250V
- 100–120V: T5A / 250V
Regulatory Marks: CE, CSA
X-Ray Source
Model: XRS 4
Type: 304534-7000-001
Serial Number: 507692
Manufacturer: Carl Zeiss Meditec AG
Energy: 50 kV
PRS Control Console
Model: PRS 500
Manufacturer: Carl Zeiss Surgical GmbH
Power Requirements: 100–240V
Frequency: 50–60 Hz
Power Consumption: 60 VA
User Terminal / CPU Workstation
Model: PRS 500 Terminal
Part Number: P3060001
Serial Number: 101080
Date of Manufacture: 2011
Operating System Label: Windows XP Embedded
Power Consumption: 100 VA
Dosimetry Equipment:
Electrometer
Manufacturer: PTW Freiburg
Model: UNIDOS E
REF: T10010
Serial Number: 000609
Ionization Chamber
Model: TN23342A
Serial Number: 003048
Manufacturer: PTW Freiburg
Additional Identified Components
• INTRABEAM Ion Chamber Shield
• INTRABEAM PDA Photodiode Array
• INTRABEAM XRS Quality Tool Set
• Centering / positioning device
• Sterile stainless instrument container
• Dosimetry cables and accessories
• Radiation warning placards
• Calibration media and documentation
Condition Summary:
Overall Cosmetic Condition
System components show signs of prior professional clinical use with expected cosmetic wear from handling, transport, and storage. Major housings, panels, and manufacturer labels appear intact.
INTRABEAM Arm and Base
The NC32 base unit and articulated arm structure appear structurally intact. Exterior panels, identification labels, and safety markings are present. Cosmetic condition consistent with institutional equipment.
PRS Control Console
Console housing and control interface appear intact with normal signs of use. Rear connectors and interlock interfaces are present.
User Terminal / CPU Workstation
Terminal workstation housing appears complete with normal cosmetic wear. Windows XP Embedded label is present on the unit.
Dosimetry Equipment
PTW UNIDOS E electrometer and TN23342A ionization chamber appear in good professional used condition and include calibration documentation.
QA Hardware and Instrumentation
Included QA accessories such as the PDA photodiode detector array, ion chamber shield, positioning fixtures, and measurement tools appear complete with typical cosmetic wear.
Documentation
System includes numerous manuals, calibration certificates, measurement reports, maintenance letters, and technical documentation.
Functionality / Testing Summary:
Testing Limitations
This is a highly specialized radiation therapy system. Complete functional verification requires specialized radiation physics equipment and trained personnel and is beyond the testing capability of a warehouse environment.
PRS Control Console
Console powers on, performs internal self-test, and progresses to the testing/operational phase.
PTW Dosimeter / Electrometer
UNIDOS E console powers on successfully and reaches the measurement interface after startup.
User Terminal / CPU
Terminal workstation powers on and boots to the Windows XP Embedded desktop environment.
Primary INTRABEAM Base / Arm System
The main INTRABEAM arm and base system were visually inspected. Full operational testing including arm movement verification, interlock testing, and radiation emission verification was not performed.
X-Ray Output
No radiation output testing was performed by our facility.
Historical Calibration & Measurement Documentation Included:
The system includes historical measurement and calibration documentation, including:
INTRABEAM XRS Output Measurement Reports
Example measurements documented:
- 50 kV / 40 µA → approximately 3.877 Gy/min
- 40 kV / 40 µA → approximately 2.885–2.907 Gy/min
Measurement certificates visible dated:
- • December 13, 2022
- • February 15, 2024
Next measurement due indicated on certificate: February 2025.
PTW Ionization Chamber Calibration
Detector: TN23342A
Calibration Date: May 10, 2023
Recommended recalibration interval: 2 years.
PTW Electrometer Calibration
Calibration documentation for the UNIDOS E electrometer is present.
Preventive Maintenance Documentation
Maintenance records reference inspections including system inspection, optical interlock verification, gold target inspection, coolant inspection, and dosimetric measurement checks.
These documents are included as historical records only.
What Is Included:
• Carl Zeiss NC32 INTRABEAM base and arm system
• PRS 500 control console
• PRS 500 user terminal / CPU workstation
• Monitor and keyboard
• XRS 4 x-ray source assembly
Dosimetry Equipment
• PTW UNIDOS E electrometer
• PTW TN23342A ionization chamber
QA Equipment
• PDA photodiode detector array
• Ion chamber shield
• XRS quality tool set
• Centering / positioning device
• Dosimetry cables
Accessories
• Sterile stainless instrument container
• Instrument holders and rails
• Radiation warning placards
Documentation
• Measurement certificates
• Calibration certificates
• Preventive maintenance documentation
• INTRABEAM manuals
• Technical documentation and calibration media
What Is Not Included:
• Spherical applicators
• Flat applicators
• Water phantom setup
• Installation or commissioning services
• Any items not shown in photos
Buyer Advisory Note:
This system is a complex radiation therapy platform requiring installation, preventive maintenance, calibration, and commissioning by qualified professionals.
While historical calibration and measurement documents are included, no current PM, calibration certification, or clinical readiness is being guaranteed by our facility.
Prior to clinical use, the buyer should arrange:
• Preventive maintenance
• Radiation output calibration
• Dosimetric verification
• Safety interlock testing
• Full system commissioning
by OEM-trained service engineers and/or licensed medical physics professionals.
Shipping Note:
This is a large, heavy, and delicate medical radiation therapy system requiring specialized freight handling.
Shipping will require palletized and/or custom crated freight transport.
Due to the size and complexity of the equipment:
• Freight shipping charges will be significant
• Additional packing and specialized handling charges may apply
• Buyer may arrange their own freight carrier
• Local pickup is available
Final shipping cost will depend on destination and freight carrier.
*Items shown in photos are the items that will ship. If it is not shown in the photos, it is not included*
Stock ID : 39155 (P.O 3034) (WH 104)
Comments:
Kindly enlarge the images and see them with zooming.
Concern or query: Ask a Question
Condition: USED, please refer to images for cosmetic condition.
- 1. Overall Condition: As seen in images - Good
- 2. Physical Condition: Please review images before bidding - Good
- 3. Working Condition: The item has been tested to power on; no detailed inspection has been performed....Will receive item as pictured……………(Offered AS IS)
- 4. “As a whole; the item has not been tested technically or professionally at our end- unable to test --- being sold STRICTLY AS IS! Biomed check is strongly recommended before clinical use!”
- 5. Completeness: (with everything seen in the pictures. Please look at all pictures and go through detail description before bidding.)