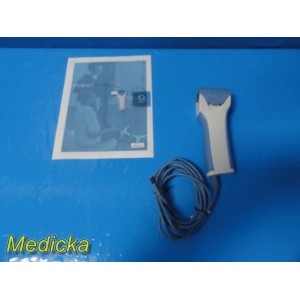
2019 Midmark IQspiro USB Sp...
2019 Midmark IQspiro USB Spirometer Ref 1-100-1225 | USB Based ~ 38499
Item specifics
- Used
- Model
- IQspiro
MPN- 1-100-1225
Product details
Manufacturer | Midmark |
Item: | 2019 Midmark IQspiro USB Spirometer REF 1-100-1225 – USB Based |
Model/Cat # | IQspiro | Ref 1-100-1225 |
Serial No. | 559011 |
Made In: | USA |
Date of Manufacture: | 2019-01-23 |
Voltage: 5V / 80mA
Short description:
This listing is for a genuine Midmark IQspiro USB Spirometer used for pulmonary function (spirometry) testing. It features an integrated USB cable connection for use with the Midmark IQ diagnostic software platform.
Reusable / Non-Sterile
Integrated USB Cable (Approx. 10 ft)
Condition Summary:
• Clean cosmetic condition
• No dents or cracks observed
• Minor surface scratches from normal handling
• USB connector and strain relief are intact
• Cable jacket shows no cuts or tears
Functionality / Testing Summary:
This device has not been tested or calibrated at our facility.
It is being sold as-is, based on visual inspection only.
Buyers should have the unit evaluated and calibrated by a qualified biomedical technician before clinical use.
What Is Included:
• Midmark IQspiro USB Spirometer
• Integrated USB Cable (Approx. 10 ft)
What Is Not Included:
• Disposable mouthpieces
• Nose clips
• Calibration syringe
• Software, licenses, or laptop
Minimum System Requirements (from Midmark spec sheet):
• Compatible with Midmark IQconnect™ Framework
• Requires Midmark diagnostic software and license
• USB connection to PC
• Windows-based operating system
(Software and license required and not included in this listing.)
Buyer Advisory Note:
Spirometry devices should be tested and calibrated prior to patient use.
No disposables or software are provided.
Shipping Note:
Approximate packed weight: 3 lbs
Ships securely packaged.
International shipping available.
*Items shown in photos are the items that will ship. If it is not shown in the photos, it is not included*
Stock ID : 38499 (P.O 2022) (SC 17)
Comments:
Kindly enlarge the images and see them with zooming.
Concern or query: Ask a Question
Condition: USED, please refer to images for cosmetic condition.
- 1. Overall Condition: As seen in images - Very Good
- 2. Physical Condition: Please review images before bidding - Very Good
- 3. Working Condition: This device has not been tested or calibrated at our facility.....will receive item as pictured…………(Offered AS IS)
- 4. “As a whole; the item has not been tested technically or professionally at our end- unable to test --- being sold STRICTLY AS IS! Biomed check is strongly recommended before clinical use!”
- 5. Completeness: (with everything seen in the pictures. Please look at all pictures and go through detail description before bidding.)